Aerobic exercise is one of the best “medicines” we have, and yet, every so often, someone collapses at a finish line, and it shakes our confidence in our so-called healthy habits (some call it an obsession).
The scientific story here is complicated. At the population level, regular exercise is strongly protective against cardiovascular disease and mortality. But when you zoom in on very high lifetime training volumes—especially in older men—the literature starts to show a more mixed picture.
The best-established example is atrial fibrillation (AFib). Long-term endurance athletes, particularly male and master athletes, show higher AFib rates than sedentary peers. That’s likely a mix of structural remodeling (bigger chambers of the heart known as the atria), autonomic shifts (more parasympathetic tone), and repeated inflammation/strain from intense exercise and racing. Ventricular arrhythmias (abnormal heart rhythms originating in the larger chambers of the heart known as the ventricles) are a different beast: they’re less common, potentially more serious, and much harder to predict. The debate in sports cardiology is whether endurance exercise can cause ventricular arrhythmias (by inducing scarring or right ventricular remodeling), or whether exercise primarily reveals an existing vulnerability (a subtle cardiac abnormality, a genetic predisposition, or a pre-existing scar) by providing a potent trigger.
Many experts describe a “perfect storm” model as an explanation. During intense exercise, you stack pro-arrhythmic ingredients (high heart rate, stress hormones, stimulants, dehydration) on top of an arrhythmia-prone heart. That could spell disaster.
But in real life, endurance athletes often look “normal” on routine screens, and the arrhythmias can be intermittent. Which raises a practical question: is there something about how we train—volume, intensity, pattern—that meaningfully changes risk? Or is this mostly about who you are and what your heart tissue looks like?

A new study tries to answer that with unusually granular data and very rigorous measurement choices:
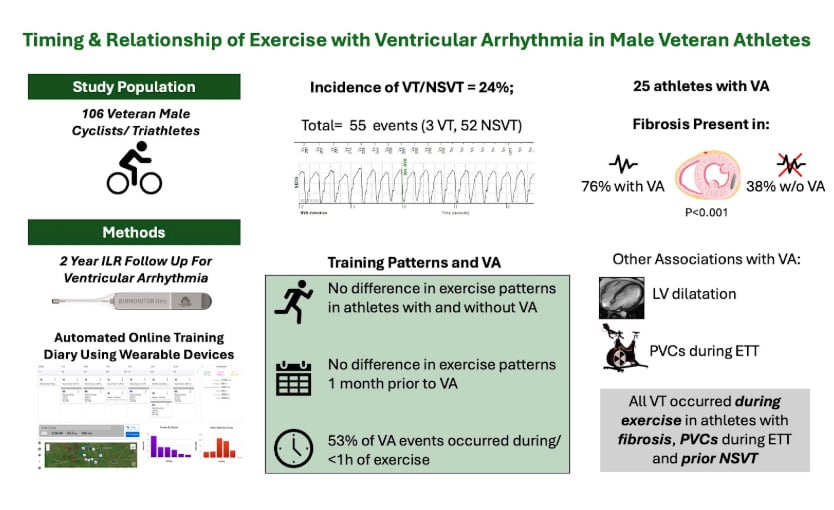
Researchers recruited 106 “healthy” veteran male endurance athletes (cyclists/triathletes), all >50 years old, with a long history of heavy training (>10 hours/week for >15 years). Importantly, they excluded individuals with known cardiovascular disease and athletes with symptoms suggestive of disease (chest pain, palpitations, shortness of breath, or fainting).
Everyone underwent a clinical examination, a 12-lead electrocardiogram (ECG) to monitor heart rhythm, cardiac magnetic resonance imaging (MRI) to detect cardiac scarring and fibrosis, and a supervised maximal exercise test to measure maximum heart rate, performance variables, and anchor training-load variables (described later).
The training load measurement is where the study gets unusually detailed. Athletes used computerized exercise-tracking devices and synchronized their workouts with an online training diary (TrainingPeaks). Training wasn’t just volume; they quantified intensity in multiple ways using frequency, duration, and distance from the recorded sessions. They also calculated Training Stress Score (TSS) as their primary training load metric.
They also broke intensity into time-in-zones (Z1–Z6), using standardized zone cutoffs relative to each athlete’s physiology:
That matters because it allows them to ask a more precise question than simply how much athletes trained. They can ask more precise questions about the distribution of training intensity.
Every participant received an implantable loop recorder—a small device placed under each athlete’s chest skin that was programmed to record a fast and arrhythmic heart rate faster than the participant’s max HR from the exercise test, for ≥8 consecutive beats. Athletes logged symptoms in a linked patient app, but crucially, events were not dependent on symptoms. Here’s how they defined ventricular arrhythmia events:
- Sustained ventricular tachycardia (VT): ≥30 seconds of ventricular rhythm at ≥100 bpm
- Non-sustained ventricular tachycardia (NSVT): ≥3 consecutive ventricular beats lasting <30 seconds
How often did arrhythmias show up?
During a follow-up period of just over 2 years (796 days), 55 ventricular arrhythmia events occurred in 25 athletes (23.5% of the participants): 52 NSVT (94.5% of all events) and 3 sustained VT (5.5% of all events)
Of the 55 events:
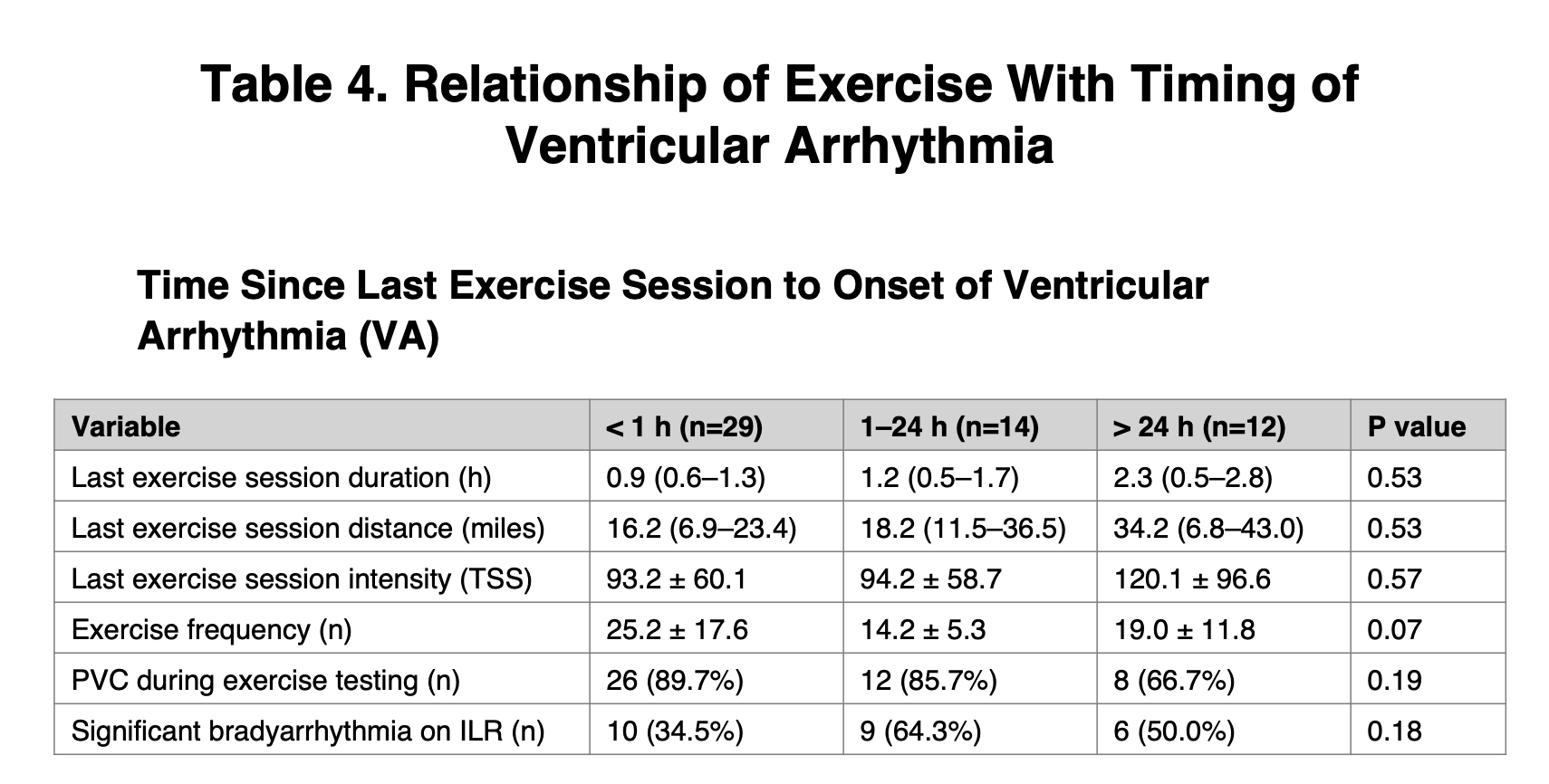
- 29 (52.7%) occurred during or within 1 hour of exercise
- 14 (25.5%) occurred 1–24 hours after exercise
- 12 (21.8%) occurred >24 hours after exercise
- 5 (9.1%) were nocturnal (occurred at night)
The incidence rate (the headline risk statistic) was 0.4 events per 1,000 hours of exercise vs 0.01 per 1,000 hours of non-exercise time. That is 28.6× higher event rate with exercise exposure.
Here’s where the study gets interesting. It identifies myocardial fibrosis as the clearest risk factor for ventricular arrhythmias in athletes. Among athletes with detectable fibrosis, 19/50 (38%) developed an arrhythmia during the follow-up period. Among those without fibrosis, only 6/56 (10.7%) developed an arrhythmia.
That’s an absolute difference of +27.3 percentage points, and a relative risk of about 3.6× higher (roughly +255% higher risk) in athletes with fibrosis.
Furthermore, all three sustained VT cases (sustained VT being the longer-lasting, more serious form) occurred during exercise in athletes with fibrosis, and each was preceded by NSVT.
Premature ventricular contractions (early heartbeats occurring during maximal exercise testing) were another clear discriminator of who would develop an arrhythmia during follow-up.
During exercise testing, 21/25 (84.0%) of athletes in the arrhythmia group experienced PVCs vs 35/74 (47.3%) in the no-arrhythmia group.
The influence of training characteristics
From the objective (TrainingPeaks/exercise device) data, we observed that most training characteristics were largely identical between groups that experienced and did not experience an arrhythmia.
- Weekly total exercise: 7.2 ± 3.1 hours (no arrhythmia) vs 7.2 ± 3.0 hours (arrhythmia)
- Weekly cycling distance: 1,17.2 ± 57.3 miles vs 1,10.0 ± 46.1 miles
- Weekly TSS: 338 (229–484) vs 366 (284–448)
- Annual cycling distance: 6,094 ± 2,979 miles vs 5,722 ± 2,400 miles
- Monthly session frequency: 14.5 (10.2–18.8) vs 16.4 (12.6–20.1)
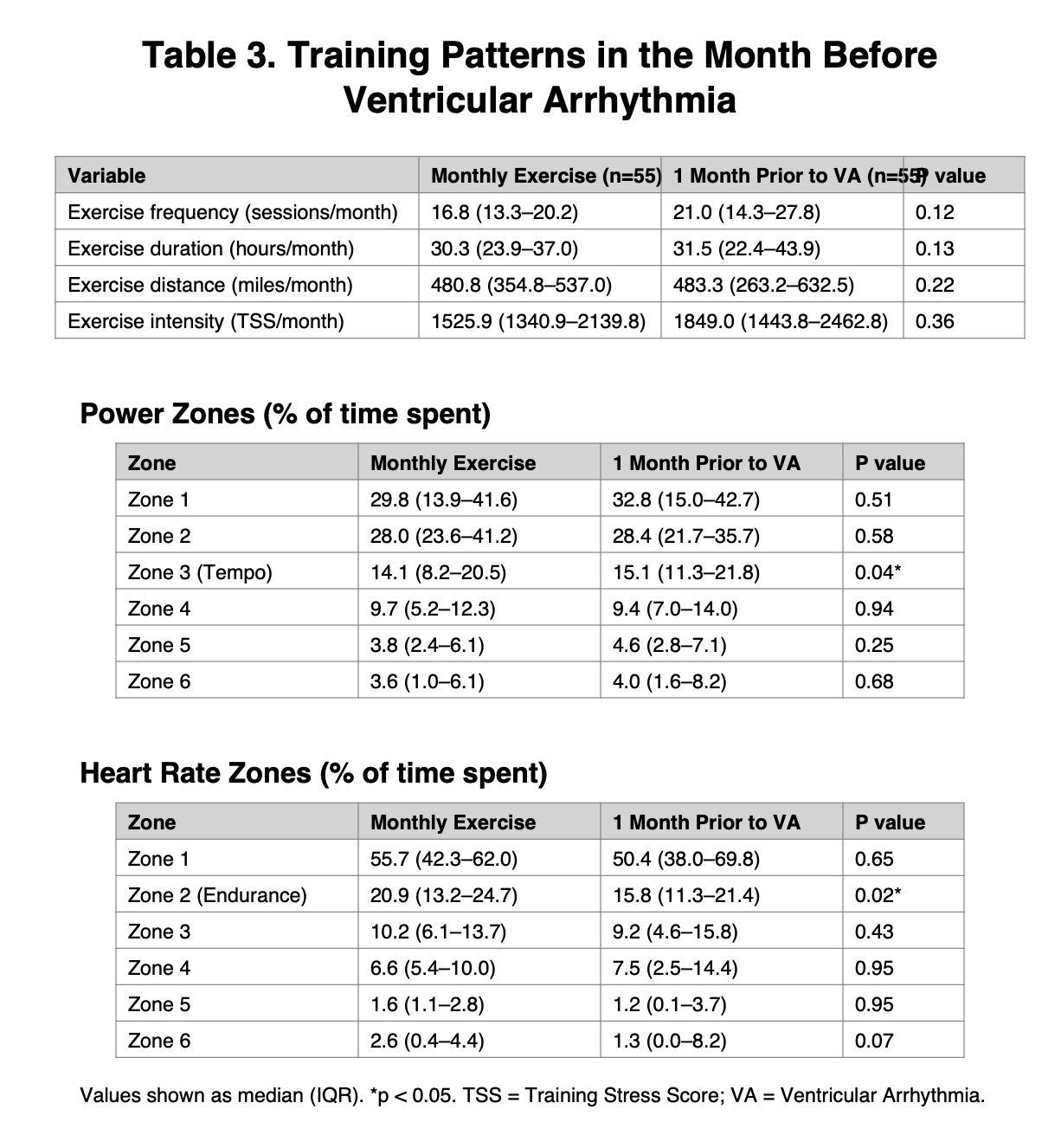
Even in the month before each arrhythmic event (within each athlete), the big training load variables weren’t much different compared to the rest of the time when they didn’t experience an arrhythmia.
- Frequency: 16.8 vs. 21.0 sessions/month
- Duration: 30.3 vs. 31.5 hours/month
- Distance: 480.8 vs. 483.3 miles/month
- TSS: 1,525.9 vs. 1,849.0 per month
The only meaningful-looking shifts were distributional (time in zones), not total load:
- Power Zone 3 (tempo): 14.1% vs. 15.1% of time
- HR Zone 2 (endurance): 20.9% vs. 15.8% of time
And when they grouped events by how soon after exercise they happened, the preceding session metrics weren’t clearly different:
- Last-session duration: 0.9 h (<1h group) vs 1.2 hours (1–24h) vs 2.3 hours (>24h)
- Last-session distance: 16.2 miles vs 18.2 miles vs 34.2 miles
- Last-session TSS (mean): 93.2 vs 94.2 vs 120.1
What is this telling us? Doing more intensity, riding for longer, or having a sharp spike in intensity doesn’t necessarily mean you’re more likely to experience an arrhythmia in the ensuing 24 hours, nor does it make you more likely to experience one sooner after your exercise session (like 1 hour after) versus later (24 hours or more after).
Other physiological differences worth noting (numbers)
A few other physiological differences that might be worth noting: Athletes who experienced arrhythmias during the study had larger left ventricular volumes, on average (about 7.5% larger), compared to athletes without arrhythmias. However, cycling functional threshold power (FTP) was similar between groups—250 W (arrhythmia) vs. 239 W (no arrhythmia).
What this means for runners
The practical message is not to train less so your heart won’t explode. Because in this cohort the athletes with arrhythmias didn’t have higher weekly hours, distance, or TSS; rather, the signal was substrate/susceptibility + trigger: myocardial fibrosis was associated with a much higher observed arrhythmia risk (fibrosis roughly tripled risk), and then acute exercise exposure clustered tightly around when events actually happened (nearly 29× higher event rate during/near exercise than away from it).
So the actionable thing for veteran endurance athletes (or those of us hoping to become them) is this: don’t ignore symptoms during exerciseexertional palpitations/lightheadedness, take abnormal heart sensations seriously, and recognize that the best “risk reduction” may be identifying whether you have an underlying risk like fibrosis, rather than trying to perfectly tweak your intensity distribution or scrutinize your training load. Because when it comes to your heart, risk might be influenced more by who you are than what you’re doing.










